Will Favorable Camzyos Results Enhance BMY's Cardiovascular Product Line?
Bristol Myers’ Cardiovascular Portfolio: Recent Developments
Bristol Myers Squibb (BMY) currently features two major cardiovascular therapies in its lineup: Eliquis and Camzyos.
Recently, the company shared encouraging findings from the SCOUT-HCM phase III trial, which assessed Camzyos (mavacamten) in adolescents aged 12 to under 18 with symptomatic obstructive hypertrophic cardiomyopathy (oHCM). This marks the first time a cardiac myosin inhibitor (CMI) has been studied in this younger patient group.
The trial achieved its main goal, demonstrating that Camzyos significantly reduced the Valsalva left ventricular outflow tract (LVOT) gradient at 28 weeks compared to placebo. This result highlights Camzyos’ ability to ease LVOT obstruction.
Additionally, Camzyos showed benefits on several secondary measures at the 28-week mark, and its safety profile was similar to that of the placebo group.
These results suggest Camzyos could become the first CMI approved for treating oHCM in adolescents.
At present, Camzyos is already authorized for use in adults with symptomatic New York Heart Association (NYHA) class II–III obstructive hypertrophic cardiomyopathy to enhance physical function and relieve symptoms.
If Camzyos receives approval for a wider range of patients, it could further drive revenue growth. In 2025, the drug generated more than $1 billion in sales, reflecting a 77% increase from the previous year.
Another key product in BMY’s cardiovascular segment is Eliquis, a blood thinner. Bristol Myers co-develops and co-markets Eliquis globally with Pfizer (PFE), and the medication remains a significant revenue driver.
However, in late 2025, BMY’s cardiovascular pipeline faced a setback when the company halted the phase III Librexia trial for milvexian, an investigational oral factor XIa (FXIa) inhibitor.
Bristol Myers and its partner Johnson & Johnson (JNJ) were testing milvexian’s safety and effectiveness when added to standard antiplatelet therapy for patients who had recently experienced an acute coronary syndrome (ACS) event.
The decision to stop the Librexia ACS study followed a planned interim review by the Independent Data Monitoring Committee (IDMC), which concluded the trial was unlikely to achieve its primary efficacy goal.
Despite this, the IDMC recommended that two other late-stage studies—Librexia AF (for atrial fibrillation) and Librexia STROKE (for secondary stroke prevention)—should continue as scheduled. Initial results from these trials are anticipated in 2026.
Competitive Landscape in Cardiovascular Treatments
In December 2025, Cytokinetics (CYTK) received FDA approval for aficamten, marketed as Myqorzo, for treating obstructive HCM in the United States. This approval marks Cytokinetics’ transition from a development-focused biotech to a commercial-stage company.
The launch of Myqorzo is a major milestone for Cytokinetics, given the significant market opportunity in oHCM.
Johnson & Johnson’s Xarelto, another Factor Xa inhibitor like Eliquis, also competes in this space. However, Xarelto is currently facing patent disputes in the U.S. JNJ co-developed Xarelto with Bayer AG.
Bristol Myers: Stock Performance, Valuation, and Analyst Estimates
So far this year, Bristol Myers’ stock has risen by 8%, outperforming the broader medical-biomedical and genetics industry, which has declined by 3.4%.
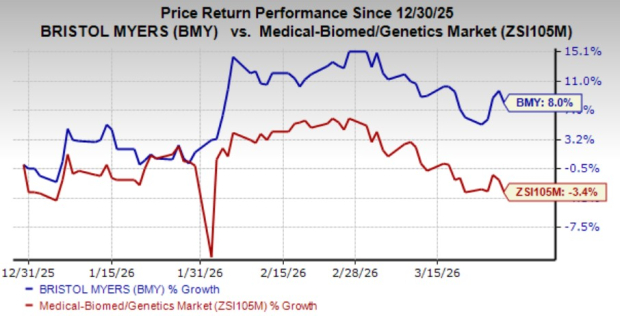
Image Source: Zacks Investment Research
From a valuation perspective, BMY trades at a lower price/earnings ratio compared to the large-cap pharmaceutical sector. The stock’s forward P/E is currently 9.41, above its historical average of 8.51 but well below the industry average of 16.74.
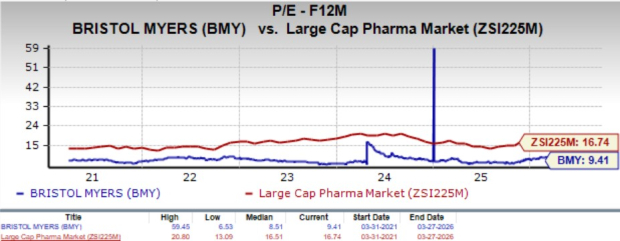
Image Source: Zacks Investment Research
Analyst consensus for BMY’s 2026 earnings per share has increased to $6.26 from $6.24 over the past month, while the 2027 estimate has risen to $6.09 from $6.05.
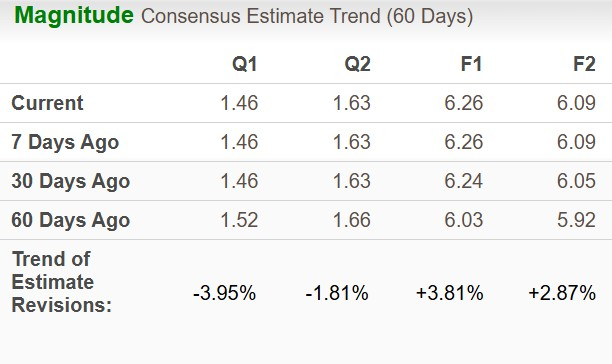
Image Source: Zacks Investment Research
Currently, BMY holds a Zacks Rank #3 (Hold).
Special Report: Capitalizing on the Next AI Boom
The upcoming wave of artificial intelligence innovation is expected to generate substantial economic value and transform many aspects of daily life. Early investors could see significant returns.
Those who invested in companies like Nvidia at the right time have already experienced impressive gains.
However, the rapid ascent of the initial group of AI stocks may soon plateau, paving the way for a new set of emerging leaders in the sector.
Zacks’ report, AI Boom 2.0: The Second Wave, highlights four lesser-known companies that could become major players in the next phase of AI growth.
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
Microsoft Stock Soars to Lead Trading Activity as AI Advancements Drive Increased Investor Interest
Nvidia Tops Market in Trading Volume as AI and Space Computing Sectors Grow
BP Token Debuts Featuring Airdrop and Share Conversion System

Meta Surges to Top Volume Spot Amid Legal Woes and AI Betting
