BioRestorative Therapies, Inc. Issues Letter to Shareholders Highlighting Major Milestones, Regulatory Alignment, Clinical Execution and Capital Strategy
MELVILLE, N.Y., March 04, 2026 (GLOBE NEWSWIRE) -- BioRestorative Therapies, Inc. (“BioRestorative”, “BRTX” or the “Company”) (NASDAQ:BRTX
To our shareholders and stakeholders,
The first quarter of 2026 marks a pivotal period for BioRestorative Therapies. We have achieved significant milestones and have strong momentum across our clinical programs, regulatory pathways, commercial initiatives, and capital strategy. We believe the Company is well-positioned at a meaningful value inflection point, supported by:
- Completion of enrollment in our fully powered Phase 2 trial of BRTX-100
- Positive Type B regulatory outcome and alignment on the BLA pathway
- Initiation of Phase 3 enabling activities
- Strengthened capital position following a $5 million public offering
- Acceleration of our commercial BioCosmeceutical strategy
- Continued development of our ThermoStem
®metabolic platform
Independently, each of these milestones is value-creating. Collectively, they represent a convergence of clinical, regulatory, and commercial momentum.
In February 2026, we completed enrollment of 99 patients across 15 U.S. sites in our prospective, randomized, double-blind, sham-controlled, single-disc Phase 2 trial of BRTX-100, our autologous hypoxically cultured mesenchymal stem cell therapy for chronic lumbar disc disease (cLDD). This study represents the largest and most rigorously designed FDA-authorized cell therapy trials conducted in cLDD. The sham-controlled design establishes a high evidentiary threshold — one consistent with registrational expectations and payer scrutiny in spine medicine. Rapid enrollment over the last 6 months across a broad network of investigators reflects both clinical demand and site engagement, reinforcing our view of the unmet need and potential commercial opportunity. It also reflects the positive momentum associated with a pristine safety profile and strong efficacy signal on blinded data. Topline results are expected following database lock and blinded statistical analysis.
Amidst completion of enrollment, we held a Type B meeting with the U.S. Food and Drug Administration, during which we confirmed alignment on our proposed Phase 3 trial design, study endpoints and assumptions, dosing strategy, sample size, and powering assumptions. Importantly, no safety concerns were raised, and our Chemistry, Manufacturing, and Controls (CMC) framework was deemed appropriate for late-stage development. Regulatory alignment at this stage significantly reduces development pathway uncertainty and allows us to initiate Phase 3 enabling activities with increased confidence. We are targeting submission of a Phase 3 IND amendment in the second half of 2026. This regulatory progress is central to our capital allocation strategy and to our long-term value-creation plan, and these achievements provide meaningful clarity on a potential accelerated Biologics License Application (BLA).
Our BioCosmeceutical platform represents a core pillar of our capital strategy and near-term revenue acceleration plan. We are expanding the distribution of products powered by our proprietary secretome-based biologics platform. The appointment of Crystal Romano as Head of Global Commercial Operations further strengthens our execution capabilities and positions us to accelerate commercialization across our cell-based portfolio. Crystal brings nearly two decades of executive leadership experience across the medical, aesthetics, and regenerative medicine sectors. This initiative is designed to generate non-dilutive revenue streams, support brand and manufacturing validation, and strategically offset development burn as we advance our clinical pipeline.
- BioCosmeceuticals sit at the intersection of skincare, aesthetics, and regenerative medicine, making them one of the fastest-growing segments in beauty and longevity, with the broader regenerative aesthetics market projected to exceed $28B by 2030. These products leverage biologically active ingredients that signal cell molecules that influence tissue repair pathways and are increasingly integrated into professional aesthetic procedures, medical-grade skincare, and at-home regenerative regimens. Clinics are rapidly adopting these solutions, with exosome treatments now used globally, signaling a clear shift: skincare is moving from cosmetic improvement to true biological performance
- BioRestorative is uniquely positioned to lead in this market by entering as a biotechnology company rather than a traditional beauty brand. As a cGMP-compliant, ISO-7 cleanroom cell manufacturing organization advancing therapeutic products under rigorous cell and tissue protocols, the Company brings fully integrated regenerative manufacturing and scientific infrastructure to a category largely driven by marketing-focused skincare brands. This foundation provides vertical control over cell sourcing, processing, quality systems, and scalable manufacturing, driving consistency, supply reliability, and long-term defensibility. BioRestorative’s therapeutic heritage also creates scientific credibility and regulatory discipline, critical differentiators in an emerging market, and enables the Company to translate regenerative science into differentiated, premium biocosmeceutical products.
- We are building a vertically integrated regenerative beauty platform engineered for scale, credibility, and premium monetization. Our three-channel strategy—global distribution partnerships, enterprise clinical adoption, and a high-margin direct-to-consumer engine—enables us to capture volume, validation, and recurring revenue simultaneously. Clinical integration drives authority, enterprise accounts provide predictable scale, and DTC extends in-office treatments into recurring at-home regimens, creating a powerful flywheel of demand and retention. The result is a scalable, defensible platform that combines biotech differentiation with beauty-level margins, positioning BioRestorative as a leader in next-generation regenerative skincare.
Our ThermoStem
Following our recent $5 million public offering, we have strengthened our balance sheet and extended our operational runway. We are focused on maintaining optionality, advancing BRTX-100 through key data readouts, and preserving leverage ahead of potential partnership discussions. Throughout this period of rapid operational progress, we have remained disciplined in our capital strategy. This financing follows a previously completed registered direct offering that included participation from healthcare investors and members of our executive leadership team, reflecting confidence in our strategy, our science, and our execution capabilities.
Our primary focus in 2026 is advancing BRTX-100 into late-stage development with regulatory clarity and operational readiness for BLA approval. This includes the initiation of Phase 3 enabling activities, the refinement of protocol elements consistent with prior FDA feedback, the confirmation of manufacturing readiness, and the submission of the Phase 3 IND amendment, targeted for the second half of 2026. These efforts are being sequenced deliberately to ensure alignment across regulatory, clinical, and manufacturing functions prior to trial initiation.
We intend to report Phase 2 topline results in early 2027 in accordance with the pre-specified statistical analysis plan. Beyond reporting the data, our objective is to evaluate treatment effect, durability, and safety outcomes to inform Phase 3 design assumptions and long-term positioning in chronic lumbar disc disease. The results, together with regulatory feedback, will guide our registrational strategy and support informed strategic decision-making.
Outside the United States, commercial applications of cell-based biologics, such as stem cells, have advanced significantly over the past several years and have continued to grow in 2026. Recently, there has been a shift towards providing access to non-FDA-approved stem cell therapies for patients suffering from numerous musculoskeletal conditions, particularly back pain. Although not covered by insurance, states such as Utah, Wyoming, Texas, and Florida have instituted state laws that allow providers to offer therapies, such as BRTX-100, to patients. We intend to seek strategic and commercial partnerships within these states, which would allow us to potentially commercialize BRTX-100 prior to full BLA approval.
Our BioCosmeceutical platform will continue to be developed with a focus on operational execution and margin discipline. We are working to expand practitioner adoption, optimize manufacturing throughput, and establish a more predictable commercial revenue base. This initiative is intended to complement our clinical development programs while leveraging our biologics manufacturing capabilities.
With respect to ThermoStem
We remain committed to disciplined capital deployment aligned with defined clinical and regulatory milestones. Our objective is to extend operational runway, manage dilution prudently, and preserve strategic flexibility. Financing, partnership, or other strategic alternatives will be evaluated in the context of long-term shareholder value and balance sheet stability. In 2026, our financial and operational decisions are guided by a clear prioritization of readiness for late-stage development of BRTX-100. Resources are being aligned to regulatory, manufacturing, and clinical preparation activities, while other initiatives will advance in a measured manner. Our objective is to maintain organizational focus as we approach the next stage of development.
We believe BioRestorative is transitioning from development-stage uncertainty toward late-stage regulatory clarity. Our focus remains on execution, data integrity, capital efficiency, and long-term shareholder value.
On behalf of our Board of Directors and the entire BioRestorative team, thank you for your continued confidence.
Sincerely,
President, Chief Executive Officer and Chairman
BioRestorative Therapies, Inc.
BioRestorative (www.biorestorative.com) develops therapeutic products using cell and tissue protocols, primarily involving adult stem cells. As described below, our two core clinical development programs relate to the treatment of disc/spine disease and metabolic disorders, and we also operate a commercial BioCosmeceutical platform:
• Disc/Spine Program (brtxDISC
• Metabolic Program (ThermoStem
• BioCosmeceuticals: We operate a commercial BioCosmeceutical platform. Our current commercial product, formulated and manufactured using our cGMP ISO-7 certified clean room, is a cell-based secretome containing exosomes, proteins and growth factors. This proprietary biologic serum has been specifically engineered by us to reduce the appearance of fine lines and wrinkles and bring forth other areas of cosmetic effectiveness. Moving forward, we also intend to explore the potential of expanding our commercial offering to include a broader family of cell-based biologic aesthetic products and therapeutics via IND-enabling studies, with the aim of pioneering FDA approvals in the emerging BioCosmeceuticals space.
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
Bitcoin’s post-Iran strike rally reignites safe-haven debate
BC-Gold-Silver
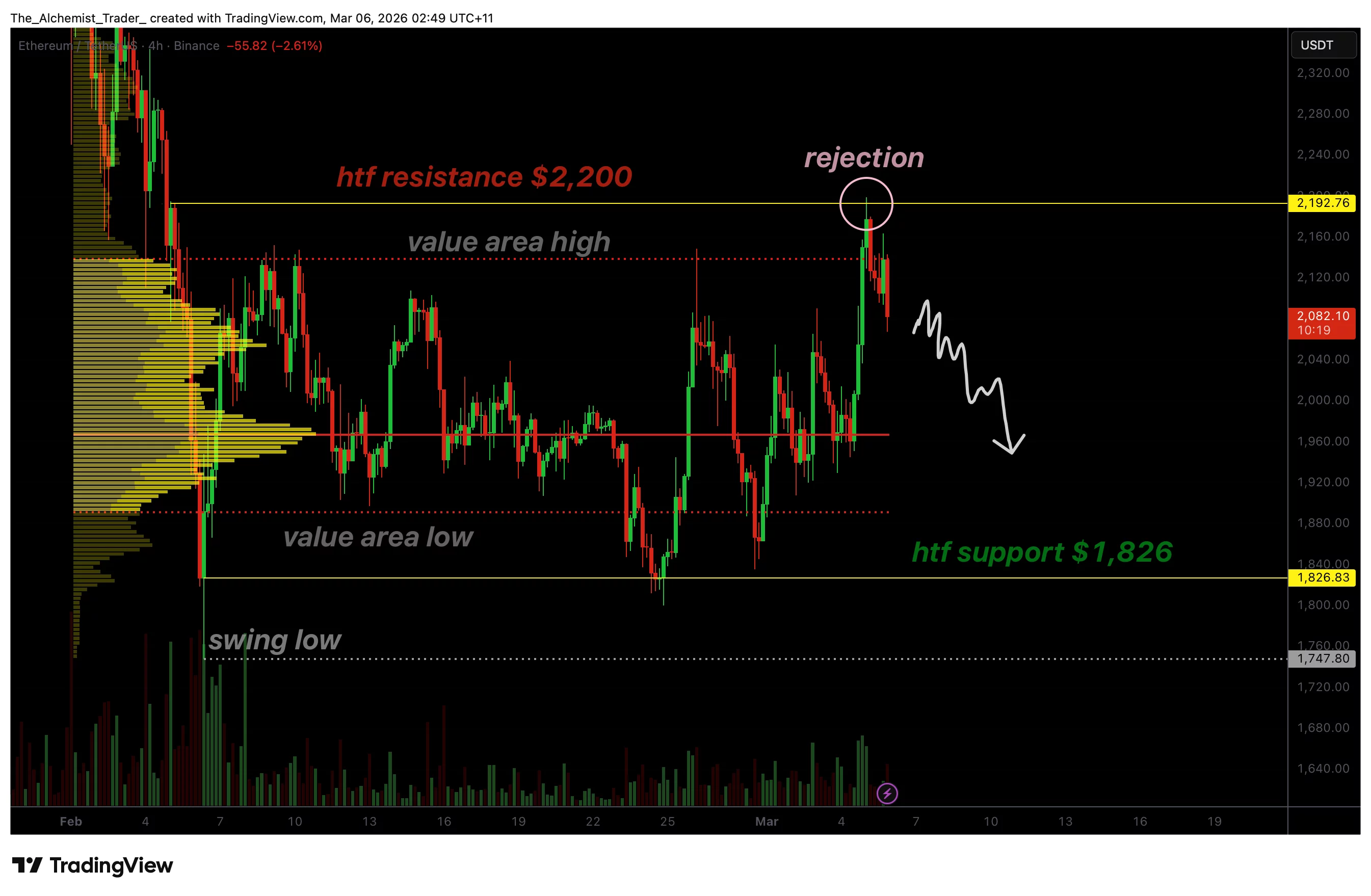
Ethereum price confirms rejection at $2,200 as downside risks build
Glencore to back Kazakh entrepreneur’s purchase of 40% stake in ERG, FT reports

