BioRestorative Therapies, Inc. to Participate in the Be+Well Beauty and Wellness Show in New York City, March 8-10, 2026
MELVILLE, N.Y., March 05, 2026 (GLOBE NEWSWIRE) -- BioRestorative Therapies, Inc. (“BioRestorative”, “BRTX” or the “Company”) (NASDAQ:BRTX)
BioRestorative will attend the event as part of its ongoing efforts to expand commercialization awareness and distribution opportunities for its BioCosmeceutical platform, which applies regenerative biologics technology to the rapidly evolving aesthetics and longevity markets. The event brings together leading companies, clinics, medspas, distributors, and wellness brands across the beauty, aesthetics, and professional skincare industries.
Crystal Romano, Head of Global Commercial Operations, together with her team, will lead the Company’s engagement at the conference, meeting with potential business and marketing partners, distributors, and practitioners to discuss product adoption, marketing collaborations, and the expanded commercial deployment of the Company’s proprietary regenerative skincare technologies.
“The Be+Well show provides an important opportunity to engage directly with industry stakeholders as we expand awareness of our BioCosmeceutical platform and highlight our unique approach, which intersects skincare, aesthetics, and regenerative medicine,” said Ms. Romano. “Our approach reflects BioRestorative’s broader regenerative medicine expertise, applying biologically active secretome components—including exosomes, growth factors, and signaling proteins—to products designed for professional aesthetics, medical-grade skincare environments, and at-home regenerative regimens. As we see in the market, clinics, medspas, and beauty and self-care venues are rapidly adopting these solutions, with exosome treatments now used globally, signaling the clear shift from cosmetic improvement to true biological performance. Regenerative technologies continue to reshape the aesthetics and longevity markets, and we believe our biologics manufacturing capabilities and scientific foundation position us to participate meaningfully in this emerging category,” Ms. Romano concluded.
BioRestorative is uniquely positioned to lead in this market by entering as a biotechnology company rather than a traditional beauty brand. As a cGMP-compliant, ISO-7 cleanroom cell manufacturing organization advancing therapeutic products under rigorous cell and tissue protocols, the Company brings fully integrated regenerative manufacturing and scientific infrastructure to a category largely driven by marketing-focused skincare brands. This foundation provides vertical control over cell sourcing, processing, quality systems, and scalable manufacturing, driving consistency, supply reliability, and long-term defensibility. BioRestorative’s therapeutic heritage also creates scientific credibility and regulatory discipline, critical differentiators in an emerging market, and enables the Company to translate regenerative science into differentiated, premium biocosmeceutical products.
“As we outlined in our recent letter to shareholders, expanding our BioCosmeceutical platform is an important component of BioRestorative’s broader strategy to generate commercial traction while advancing our clinical pipeline,” said Lance Alstodt, President, BioRestorative Chief Executive Officer and Chairman. “Our highly differentiated, vertically integrated regenerative beauty platform is engineered for scale, credibility, and premium monetization. In addition, we hope that global distribution partnerships, enterprise clinical adoption, and a high-margin direct-to-consumer engine will enable us to capture volume, validation, and recurring revenue simultaneously. This scalable, defensible platform combines biotech differentiation with beauty-level margins, positioning BioRestorative to possibly emerge as a leading company in next-generation regenerative skincare. Events such as Be+Well allow us to engage directly with the clinics, practitioners, and distribution partners that are shaping the future of regenerative aesthetics. We look forward to deepening relationships within this community and to providing updates on our progress as our commercialization efforts continue to develop.”
For over a century, the International Beauty Show (IBS) and International Esthetic, Cosmetic & Spa Conference (IECSC) have been where beauty, hair, esthetics, cosmetics, spa, and wellness professionals have come to perfect their craft, discover top brands from over 600+ Exhibitors and get world-class education in 180+ classes. Now, both shows unite under one powerful brand: Be+Well | Beauty and Wellness Show.
BioRestorative (www.biorestorative.com) develops therapeutic products using cell and tissue protocols, primarily involving adult stem cells. As described below, our two core clinical development programs relate to the treatment of disc/spine disease and metabolic disorders, and we also operate a commercial BioCosmeceutical platform:
- Disc/Spine Program (brtxDISC
™): Our lead cell therapy candidate, BRTX-100, is a product formulated from autologous (or a person’s own) cultured mesenchymal stem cells collected from the patient’s bone marrow. We intend that the product will be used for the non-surgical treatment of painful lumbosacral disc disorders or as a complementary therapeutic to a surgical procedure. The BRTX-100 production process utilizes proprietary technology and involves collecting a patient’s bone marrow, isolating and culturing stem cells from the bone marrow and cryopreserving the cells. In an outpatient procedure, BRTX-100 is to be injected by a physician into the patient’s damaged disc. The treatment is intended for patients whose pain has not been alleviated by non-invasive procedures and who potentially face the prospect of surgery. We have commenced a Phase 2 clinical trial using BRTX-100 to treat chronic lower back pain arising from degenerative disc disease. We have also obtained U.S. Food and Drug Administration (“FDA”) Investigational New Drug (“IND”) clearance to evaluate BRTX-100 in the treatment of chronic cervical discogenic pain
- Metabolic Program (ThermoStem
®): We are developing cell-based therapy candidates to target obesity and metabolic disorders using brown adipose (fat) derived stem cells (“BADSC”) to generate brown adipose tissue (“BAT”), as well as exosomes secreted by BADSC. BAT is intended to mimic naturally occurring brown adipose depots that regulate metabolic homeostasis in humans. Initial preclinical research indicates that increased amounts of brown fat in animals may be responsible for additional caloric burning as well as reduced glucose and lipid levels. Researchers have found that people with higher levels of brown fat may have a reduced risk for obesity and diabetes. BADSC secreted exosomes may also impact weight loss
- BioCosmeceuticals: We operate a commercial BioCosmeceutical platform. Our current commercial products are formulated and manufactured in our cGMP, ISO-7 certified clean room facility. Each product features a cell-based secretome enriched with exosomes, proteins, growth factors, peptides, and other carefully selected active ingredients. This proprietary biologic portfolio has been thoughtfully engineered to support skin health and longevity while addressing visible signs of aging and enhancing overall cosmetic performance. Moving forward, we also intend to explore the potential of expanding our commercial offering to include a broader family of cell-based biologic aesthetic products and therapeutics via IND-enabling studies, with the aim of pioneering FDA approvals in the emerging BioCosmeceuticals space
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
Bitcoin’s post-Iran strike rally reignites safe-haven debate
PDAC VIDEO : Canada unveils C$1.5B mine-to-market fund
BC-Gold-Silver
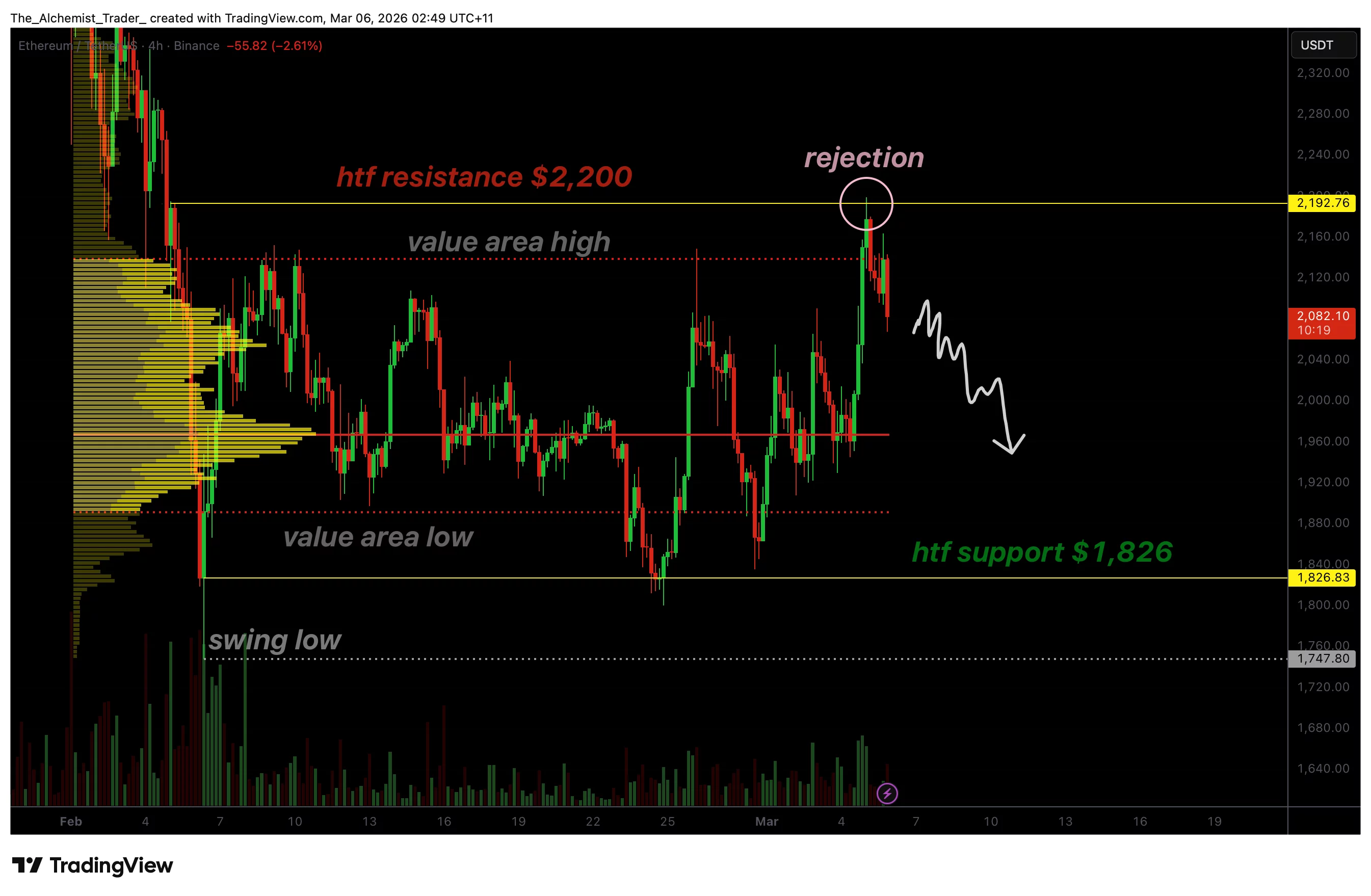
Ethereum price confirms rejection at $2,200 as downside risks build